Immunohistochemistry is a technique used to visualize the localization and relative abundance of specific antigens (proteins) within tissue sections, providing critical spatial context in their native histological environment. A specific primary antibody binds to its target antigen in a tissue section. This binding is then visualized using a detection system (typically enzyme-based, e.g., HRP or AP), which produces a colored precipitate at the site of antigen-antibody complex formation. Counterstaining provides tissue architecture context.
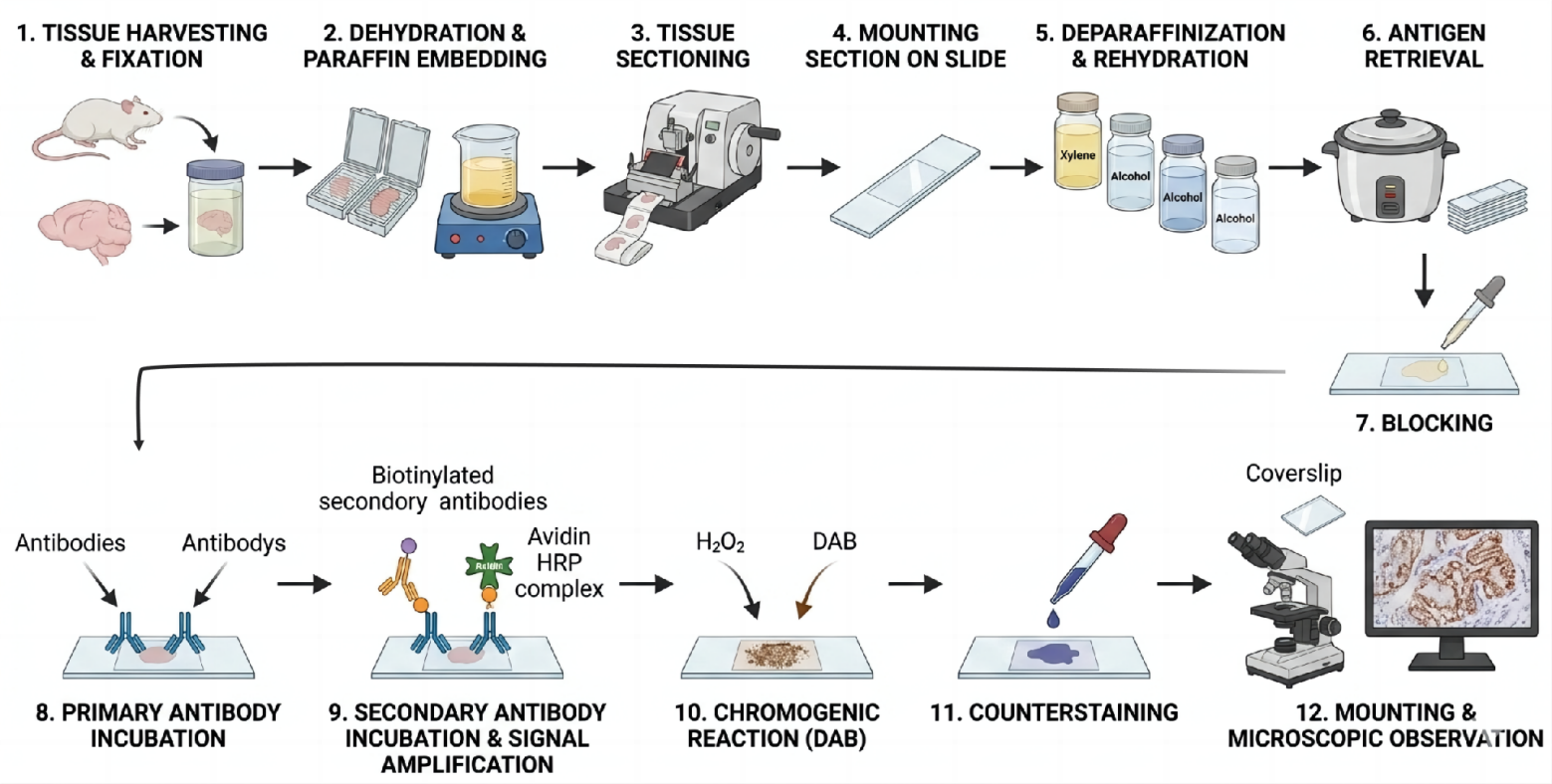
(Workflow Overview)
Stage 1: Sample Preparation & Sectioning
Performed on ice or at 4°C with pre-chilled buffers.
1.1 Tissue Fixation
Goal: Preserve tissue morphology and prevent antigen degradation.
Common Method: Neutral Buffered Formalin (NBF), 24-48 hours at room temperature.
1.2 Processing & Embedding
Paraffin-Embedded (FFPE) Tissue: Dehydrate, clear, and infiltrate tissue with paraffin wax. Embed in a paraffin block. This is the most common format.
Frozen Tissue: Embed tissue in Optimal Cutting Temperature (OCT) compound, rapidly freeze, and store at -80°C. Preserves antigens sensitive to formalin and processing but offers poorer morphology.
1.3 Sectioning
1.3.1 Cut thin sections (3-5 µm) using a microtome (paraffin) or cryostat (frozen).
1.3.2 Mount sections on positively charged or adhesive glass slides.
1.3.3 Dry slides: Paraffin sections at 37°C overnight; frozen sections at room temperature for 30-60 minutes.
Stage 2: Deparaffinization & Rehydration (For FFPE Sections Only)
2.1 Bake slides at 60°C for 20-60 minutes to melt paraffin and improve adhesion.
2.2 Deparaffinize in fresh xylene (or xylene substitute):
Xylene I: 10 minutes
Xylene II: 10 minutes
2.3 Rehydrate through graded alcohols to water:
100% Ethanol I: 5 minutes
100% Ethanol II: 5 minutes
95% Ethanol: 3 minutes
70% Ethanol: 3 minutes
Distilled Water (dH₂O): 5 minutes
Proceed immediately to antigen retrieval.
Stage 3: Antigen Retrieval (Critical Step)
To reverse formaldehyde-induced cross-linking (masking) of epitopes and restore antibody accessibility.
3.1 Choose Method
3.1.1 Heat-Induced Epitope Retrieval (HIER): Most common and effective.
Buffer: Citrate buffer (pH 6.0), Tris-EDTA (pH 9.0), or commercial retrieval solutions. Buffer choice is antigen-dependent.
Equipment: Pressure cooker, microwave, steamer, or water bath.
General Protocol (Pressure Cooker Example):
3.1.1.1 Place slides in a slide holder in pre-heated retrieval buffer.
3.1.1.2 Bring to full pressure (or ~95-100°C in other methods).
3.1.1.3 Incubate for 10-25 minutes (optimize for each antigen).
3.1.1.4 Cool naturally in the buffer for 20-30 minutes to room temperature.
3.1.2 Proteolytic-Induced Epitope Retrieval (PIER): For some antigens. Use proteinase K, trypsin, or pepsin. Incubate at 37°C for 5-30 minutes. Less common, can damage morphology.
3.2 Post-Retrieval
3.2.1 Wash slides in running dH₂O for 5 minutes.
3.2.2 Transfer to Wash Buffer (typically Tris-Buffered Saline, TBS, or Phosphate-Buffered Saline, PBS).
Stage 4: Immunostaining Procedure
4.1 Stringent Washing (Critical for Low Background)
4.1.1 Incubate slides with 3% Hydrogen Peroxide (H₂O₂) in methanol or buffer for 10-15 minutes at RT to quench endogenous peroxidase activity, especially in red blood cells and myeloid cells.
4.1.2 Wash 2 x 5 minutes in Wash Buffer.
4.2 Protein Blocking
4.2.1 Apply enough protein blocking serum (e.g., 5-10% normal serum from the species of the secondary antibody) or a commercial protein block to cover the tissue.
4.2.2 Incubate for 30-60 minutes at RT to block non-specific binding sites.
4.2.3 Do NOT wash. Tap off excess block.
4.3 Primary Antibody Incubation
4.3.1 Apply optimally diluted primary antibody in antibody diluent (often the same as the blocking serum or a commercial diluent).
4.3.2 Incubation Conditions (choose one).
Room Temperature: 1-2 hours.
4°C Overnight: Recommended for best specificity and signal, especially for low-abundance targets. Cover chamber to prevent condensation.
4.3.3 Wash 3 x 5 minutes in Wash Buffer with gentle agitation.
4.4 Secondary Antibody & Detection System
4.4.1 Apply the appropriate labeled secondary antibody or detection kit (e.g., Biotin-Streptavidin systems or polymer-based HRP/AP systems). Polymer systems (no biotin) are now standard due to higher sensitivity and lower background.
4.4.2 Incubate for 30-60 minutes at RT.
4.4.3 Wash 3 x 5 minutes in Wash Buffer.
4.5 Chromogen Development (Color Reaction)
4.5.1 Prepare chromogen substrate just before use (e.g., DAB for HRP, produces a brown precipitate; Fast Red for AP, produces a red precipitate).
4.5.2 Apply substrate to tissue and monitor development under a microscope (usually 30 seconds to 10 minutes).
4.5.3 Stop the reaction by immersing slides in dH₂O as soon as desired intensity is reached. Do not over-develop.
Stage 5: Counterstaining, Dehydration & Mounting
5.1 Counterstain
5.1.1 Immerse slides in Hematoxylin (nuclear stain) for 30-60 seconds.
5.1.2 "Blue" the sections by rinsing in running tap water or a weak ammonia solution for 5-10 minutes (turns nuclei blue).
5.2 Dehydration & Clearing (For organic mounting media)
5.2.1 Dehydrate quickly through graded alcohols: 70% EtOH → 95% EtOH → 100% EtOH I → 100% EtOH II, 30-60 seconds each.
5.2.2 Clear in xylene or xylene substitute: 2 x 5 minutes.
5.3 Mounting
5.3.1 Apply a drop of permanent, non-aqueous mounting medium (e.g., resin-based).
5.3.2 Gently lower a coverslip, avoiding air bubbles.
5.3.2 Allow to cure flat in the dark overnight.
5.4 Analysis
5.4.1 Observe and image slides using a brightfield microscope.
5.4.2 Score staining based on intensity (0, 1+, 2+, 3+) and distribution (percentage of positive cells).