A specific antibody against the target protein is immobilized on a solid support (e.g., Protein A/G magnetic or agarose beads). The antibody-bound beads are then incubated with the cell lysate, allowing the antibody to capture the target protein and its interacting partners. Non-specifically bound contaminants are removed through rigorous washing. Finally, the captured protein(s) are eluted and analyzed.
Immunoprecipitation (IP): To isolate and enrich a specific protein from a complex cellular or tissue lysate using the specific binding of an antibody to its target antigen.
Co-Immunoprecipitation (Co-IP): An extension of IP used to identify physiologically interacting protein partners ("prey") of a target protein ("bait"). The principle is that if proteins A and B are associated in the cell, then immunoprecipitating A with an anti-A antibody will also co-precipitate B.
The entire procedure can be divided into five sequential stages:
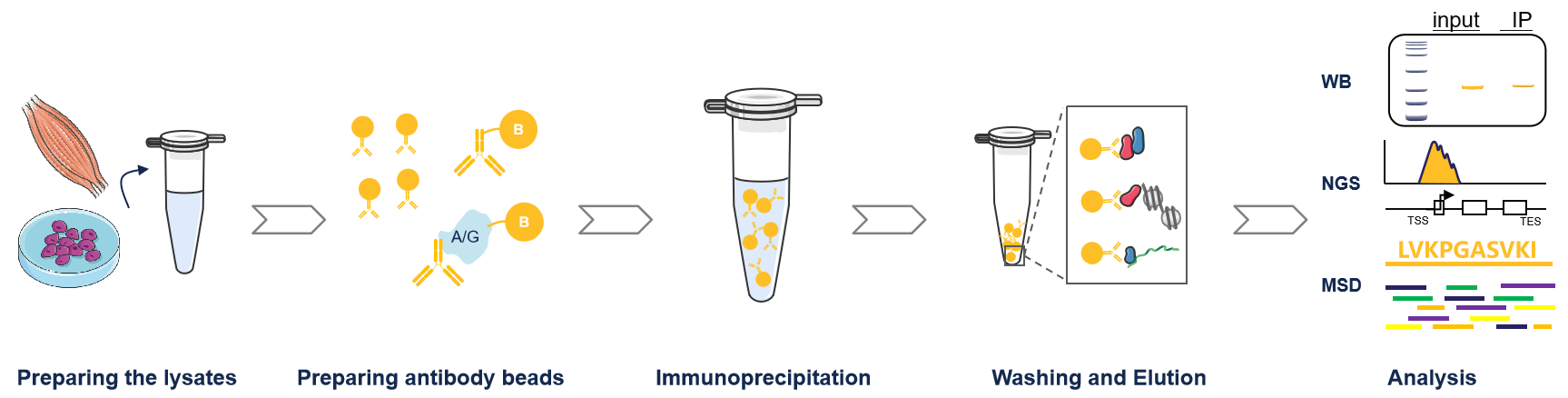
(Workflow Overview)
Stage 1: Preparation of Cell Lysate
Performed on ice or at 4°C with pre-chilled buffers.
1.1 Cell Harvesting & Lysis
1.1.1 Wash cells (adherent or suspension) with cold PBS.
1.1.2 Lyse cells in an appropriate non-denaturing IP Lysis Buffer.
Key Buffer Components:
Mild Detergent: 0.5-1% NP-40, Triton X-100, or CHAPS to solubilize proteins while preserving native interactions.
Salt: 150 mM NaCl to mimic physiological ionic strength.
Buffer: 25-50 mM Tris or HEPES (pH 7.4-7.6).
Essential Inhibitors: Add fresh protease and phosphatase inhibitor cocktails.
1.1.3 Incubate lysate on ice for 20-30 min with occasional vortexing.
1.2 Clarification
1.2.1 Centrifuge the lysate at ≥12,000 x g for 15 min at 4°C.
1.2.2 Carefully transfer the supernatant (cleared lysate) to a new tube. Discard the pellet containing insoluble debris.
1.2.3 (Optional but Recommended) Perform a pre-clearing step: Incubate the cleared lysate with bare beads (e.g., 20-50 µL bead slurry) for 30-60 min at 4°C to remove proteins that bind non-specifically to the beads. 1.2.4 Remove beads by centrifugation or magnet.
1.3 Protein Quantification
1.3.1 Measure the protein concentration of the cleared lysate using a compatible assay (e.g., BCA assay).
1.3.2 Use an aliquot (typically 0.5-2 mg total protein) for each IP reaction.
Stage 2: Antibody-Bead Preparation
2.1 Bead Selection
Protein A/G Beads: Most common. Bind to the Fc region of antibodies. Choose A, G, or A/G based on the host species and subclass of your antibody.
Magnetic vs. Agarose: Magnetic beads allow easier washing; agarose beads are traditional and cost-effective.
2.2 Bead Preparation
2.2.1 Gently resuspend the bead slurry.
2.2.2 For each IP, transfer 20-50 µL of bead slurry to a tube.
2.2.3 Wash beads 2-3 times with 1 mL of cold IP Lysis Buffer (or PBS) by gentle centrifugation (or magnet separation) to remove storage solution.
2.3 Antibody Coupling (Two Main Strategies)
Direct Method (Pre-coupled): Incubate the washed beads with 1-5 µg of the specific IP-grade antibody in 500 µL lysis buffer for 1-2 hours at 4°C on a rotator. Wash beads twice to remove unbound antibody.
Indirect Method (In-solution): First, incubate the lysate with the antibody (1-5 µg) for 1-2 hours or overnight at 4°C to form the immune complex. Then, add the pre-washed beads and incubate for another 1-2 hours to capture the complex. This method often provides higher capture efficiency.
Stage 3:Immunoprecipitation
3.1 Incubation
3.1.1 Combine the prepared antibody-bead complex (from the direct method) or the antibody-lysate mixture (from the indirect method) with the total cell lysate.
3.1.2 Incubate on a rotator or end-over-end mixer at 4°C for 2-4 hours or overnight for maximum capture.
Stage 4: Washing & Elution
4.1 Stringent Washing (Critical for Low Background)
4.1.1 Pellet the beads by brief, low-speed centrifugation (or magnet) and carefully aspirate the supernatant. Do not disturb the bead pellet.
4.1.2 Wash the bead-immune complex 3-5 times with 1 mL of cold IP Lysis Buffer. Invert tube gently 5-10 times per wash.
4.1.3 For the final wash, use a low-salt or no-salt buffer (e.g., 50 mM Tris-HCl, pH 7.5) to reduce non-ionic interactions. Remove all traces of wash buffer.
4.2 Elution
Use a low-pH glycine buffer (pH 2.5-3.0) or a high-pH buffer (e.g., 100 mM triethylamine), followed by immediate neutralization.
Stage 5: Analysis
Co-IP Analysis: Subject the eluted samples to SDS-PAGE and Western Blot.
Probe the blot first with an antibody against the co-precipitated protein ("prey") to confirm the interaction.
Re-probe the same blot with an antibody against the primary target ("bait") to confirm successful IP.
Downstream Applications: Eluted proteins can also be used for mass spectrometry, kinase assays, etc.